
Thermal oxidation of fossil fuels or other sulfur containing material generates sulfur dioxide, SO2. Petroleum refineries, secondary lead smelters, paper and pulp manufacturers, geothermal power generators, waste incinerators, and mineral processors are the primary emitters of SO2.
SO2 contributes to respiratory illness and aggravates existing heart and lung conditions. It contributes to acid rain, damaging vegetation, sensitive ecosystems, and waterways. It is one of the six common criteria pollutants. Criteria pollutants are subject to primary and secondary National Ambient Air Quality Standards (NAAQS) under the federal Clean Air Act. Primary standards prevent adverse effects on human health.
Packed bed absorbers are a common wet scrubber technology for removing SO2. Absorbers use sodium hydroxide (NaOH), often referred to as caustic, or soda ash (Na2CO3) to neutralize SO2. Relative to other air pollution control technologies, packed bed absorbers achieve high removal efficiency, possess a low capital cost, are highly automated, and require minimal maintenance with high reliability.
When absorbed into water, SO2 solubilizes to sulfite (SO3). SO3 requires further oxidation to stabilize in water. If left unoxidized, SO3 increases the chemical oxygen demand of the wastewater and can convert back to SO2 resulting in toxic offgas.
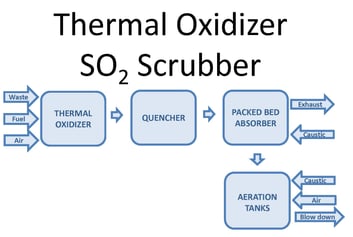
One way to oxidize the wastewater is through forced oxidation. This oxidizes sulfite (SO3) reaction products to sulfate (SO4). Forced oxidation can increase the size, complexity, and operating cost of the system. The PFD image below shows a block diagram of a thermal oxidizer SO2 scrubber with forced oxidation. Waste, fuel, and air combust in a thermal oxidizer. Sulfur compounds in the waste oxidize to SO2. After the thermal oxidizer an evaporative quencher cools the gas to its saturation temperature, typically 180°F or lower. The quencher sprays water into the gas, cooling it. Some of the water evaporates, increasing the gas water content. The gas then enters a packed bed absorber. Packing provides mass transfer to facilitate absorption of SO2 in the gas into the recirculated water. Caustic in the recirculated water reacts with dissolved SO2 by the reaction shown below:
SO2 + 2NaOH -> Na2SO3 + H2O
The reaction occurs at a pH near neutral. Excess water from the quencher and packed bed collects in the scrubber sump.
When the process requires SO3 oxidation or stabilization, aeration is integrated into the system. Caustic and air inject into the sump. Oxygen in the air oxidizes SO3. An aeration diffuser assembly promotes the transfer of oxygen into the water to facilitate the oxidation reaction.
2SO32- (aq) + O2 (g) -> 2SO42- (aq)
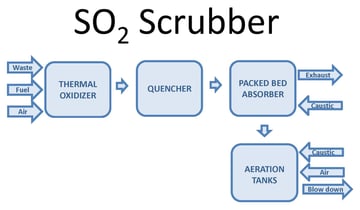 The oxidation reaction is very fast. The limiting step is dissolving oxygen into the water to allow SO3 oxidation to occur. In the case of a low sulfur load, aeration can occur in the sump with little impact on the scrubber size. In the case of a high sulfur load, the scrubber sump requires substantial modification to sufficiently oxidize the SO3. For excessive sulfur loads, oxidation may need to take place in separate oxidation tanks.
The oxidation reaction is very fast. The limiting step is dissolving oxygen into the water to allow SO3 oxidation to occur. In the case of a low sulfur load, aeration can occur in the sump with little impact on the scrubber size. In the case of a high sulfur load, the scrubber sump requires substantial modification to sufficiently oxidize the SO3. For excessive sulfur loads, oxidation may need to take place in separate oxidation tanks.
It should be noted that forced oxidation is uncommon. Most industrial SO2 packed bed absorbers don’t require forced oxidation. High sulfur load packed bed absorbers are also uncommon. For high sulfur loads, the operating cost of sodium-based reagents make higher capital cost alternatives more palatable. Other options include a limestone spray tower, a dual alkali scrubber, or a lime injection bag house. Each alternative is substantially more capital cost, more complex to operate, and require higher maintenance. When pursuing a forced oxidation SO2 absorber, it’s important to select a vendor capable of properly sizing and designing equipment for both SO2 absorption and SO2 oxidation.
Click on the link below to download SO2 scrubber literature.


 A catalyst production facility operates a calciner that generates a small stream of hot dirty gas. The exhaust is cleaned using a particulate/SO
A catalyst production facility operates a calciner that generates a small stream of hot dirty gas. The exhaust is cleaned using a particulate/SO
 The customer selected
The customer selected


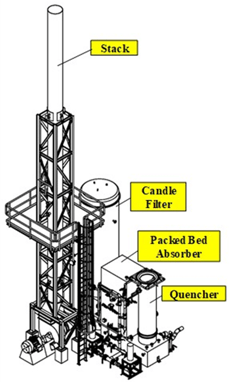 Waste oil is recycled and refined into low sulfur marine diesel and other industrial fuels at West Coast refineries. Waste gas is sent to thermal oxidizers for volatile organic compound (VOC) destruction. Sulfur compounds in the waste gas are oxidized to SO2 and removed by a packed bed scrubber. A fraction of SO2 converts to sulfur trioxide (SO3) before entering the scrubber. SO3 further converts to sulfuric acid (H2SO4) and generates a submicron liquid mist upon quenching the gas. New ground level pollutant regulations require removal of sulfuric acid mist before exhausting the flue gas to atmosphere. A multi-pollutant solution is needed to remove both SO2 and sulfuric acid mist.
Waste oil is recycled and refined into low sulfur marine diesel and other industrial fuels at West Coast refineries. Waste gas is sent to thermal oxidizers for volatile organic compound (VOC) destruction. Sulfur compounds in the waste gas are oxidized to SO2 and removed by a packed bed scrubber. A fraction of SO2 converts to sulfur trioxide (SO3) before entering the scrubber. SO3 further converts to sulfuric acid (H2SO4) and generates a submicron liquid mist upon quenching the gas. New ground level pollutant regulations require removal of sulfuric acid mist before exhausting the flue gas to atmosphere. A multi-pollutant solution is needed to remove both SO2 and sulfuric acid mist. 

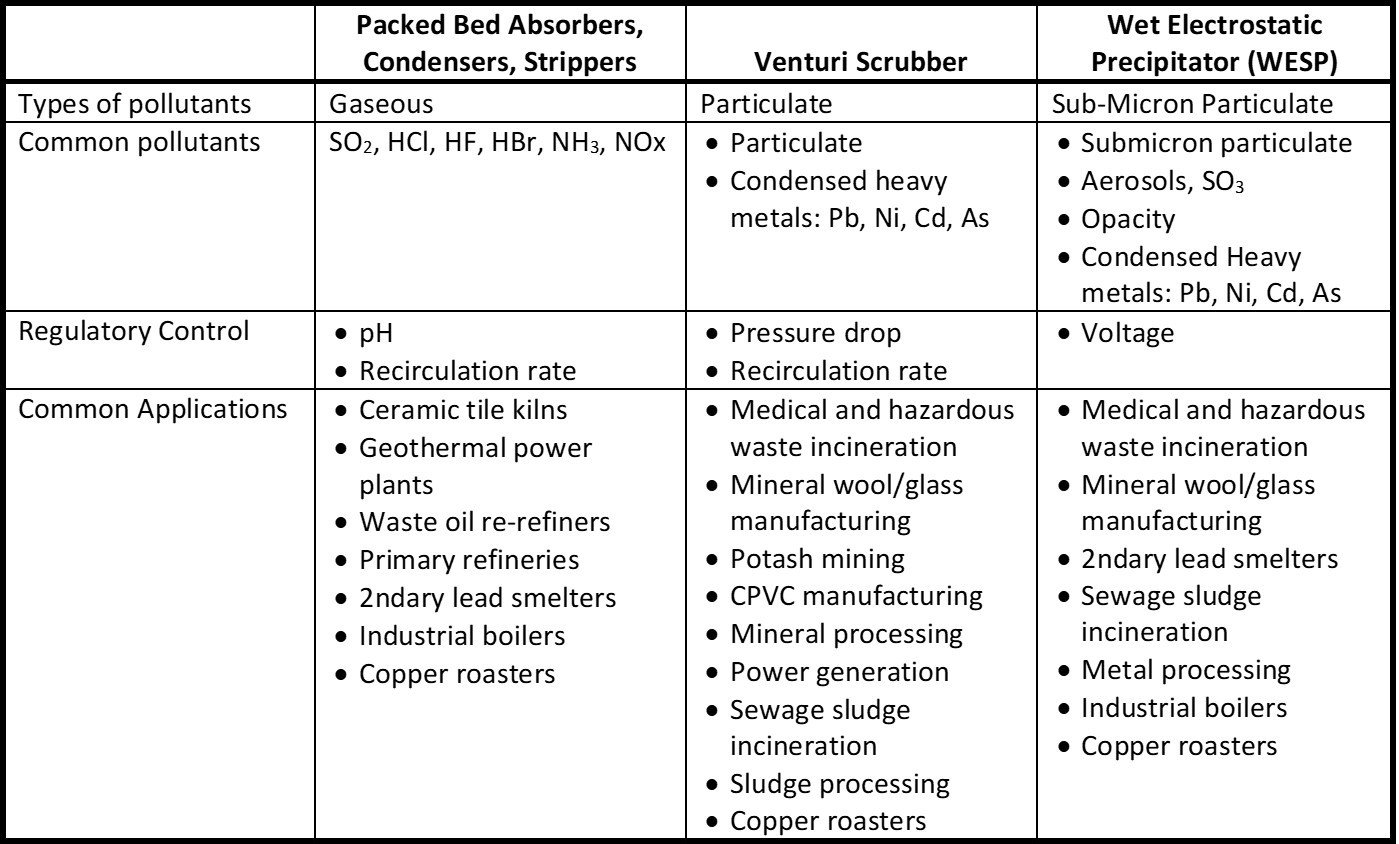
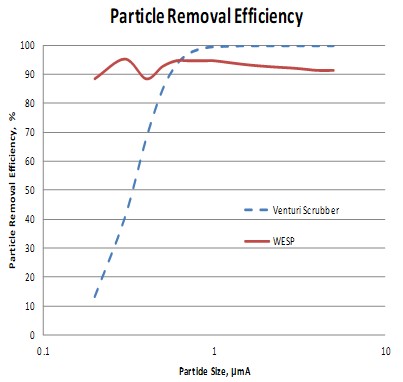
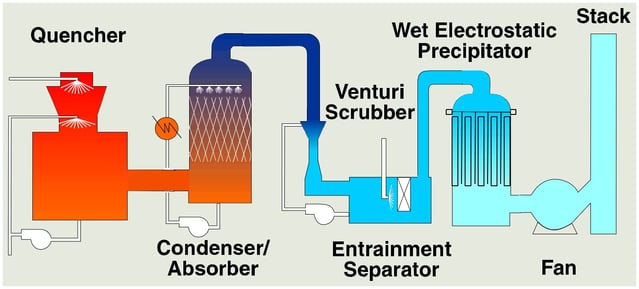


 The thermal oxidizers need a wet scrubber to neutralize and remove SO2. Flue gas entering the scrubber contain some sulfur trioxide (SO3) which is converted to sulfuric acid (H2SO4) in the quencher. Sulfuric acid is a submicron liquid aerosol that passes through the downstream packed bed absorber. Some facilities are now being regulated for H2SO4. This paper evaluates and compares candle filters versus wet electrostatic precipitators (WESP’s) for H2SO4 removal in these applications.
The thermal oxidizers need a wet scrubber to neutralize and remove SO2. Flue gas entering the scrubber contain some sulfur trioxide (SO3) which is converted to sulfuric acid (H2SO4) in the quencher. Sulfuric acid is a submicron liquid aerosol that passes through the downstream packed bed absorber. Some facilities are now being regulated for H2SO4. This paper evaluates and compares candle filters versus wet electrostatic precipitators (WESP’s) for H2SO4 removal in these applications. Waste water treatment facilities operating sewage sludge incinerators (SSI) can reduce sludge volume and disposal costs by combusting dewatered sewage sludge. Emissions are regulated by the US EPA Maximum Available Control Technology (MACT) standard 40 CFR Part 60 and 62 to control particulate, lead (Pb), cadmium (Cd), SO2, HCl, dioxins/furans, and mercury (Hg). Many SSI’s need a control device specifically for mercury. This paper evaluates two mercury control technologies: sulfur‐impregnated activated carbon and Gore sorbent polymer catalyst (SPC) modules. Several facilities have used sulfur-impregnated activated carbon but safety issues have arisen due to fires which have shut down some systems. The Gore SPC modules are a relatively new technology with at least seven installations. A comparison is made of capital cost, operating cost, mercury removal efficiency, fire and performance risks based on incineration of 3,000 lbs/hr of sewage sludge. Finally, an overview is provided for an Envitech SPC mercury control scrubber operating at one facility.
Waste water treatment facilities operating sewage sludge incinerators (SSI) can reduce sludge volume and disposal costs by combusting dewatered sewage sludge. Emissions are regulated by the US EPA Maximum Available Control Technology (MACT) standard 40 CFR Part 60 and 62 to control particulate, lead (Pb), cadmium (Cd), SO2, HCl, dioxins/furans, and mercury (Hg). Many SSI’s need a control device specifically for mercury. This paper evaluates two mercury control technologies: sulfur‐impregnated activated carbon and Gore sorbent polymer catalyst (SPC) modules. Several facilities have used sulfur-impregnated activated carbon but safety issues have arisen due to fires which have shut down some systems. The Gore SPC modules are a relatively new technology with at least seven installations. A comparison is made of capital cost, operating cost, mercury removal efficiency, fire and performance risks based on incineration of 3,000 lbs/hr of sewage sludge. Finally, an overview is provided for an Envitech SPC mercury control scrubber operating at one facility.
 Hazardous Waste Combustors (
Hazardous Waste Combustors (

 previously blogged about include refinery sulfur recovery unit tail gas treatment units (
previously blogged about include refinery sulfur recovery unit tail gas treatment units (